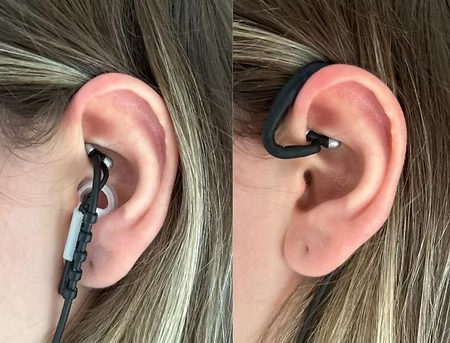
Discover why tVNS® is the preferred device for research on transcutaneous vagus nerve stimulation.
With its precise cymba concha stimulation and wireless parameter flexibility, tVNS offers cutting-edge technology and unparalleled flexibility to explore new therapeutic protocols.
Approved under the EU-MDR and backed by CB reports, our tVNS device ensures adherence to the highest standards of safety and efficacy in medical research.